ApneBoot
ApneBoot is a wearable device to alert for bradycardia and hypoxia in newborns.
Key Facts
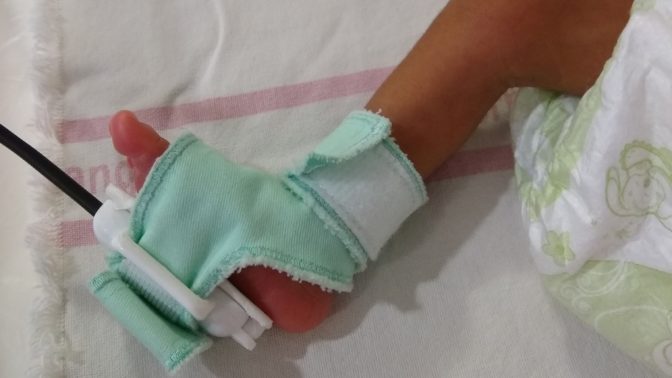
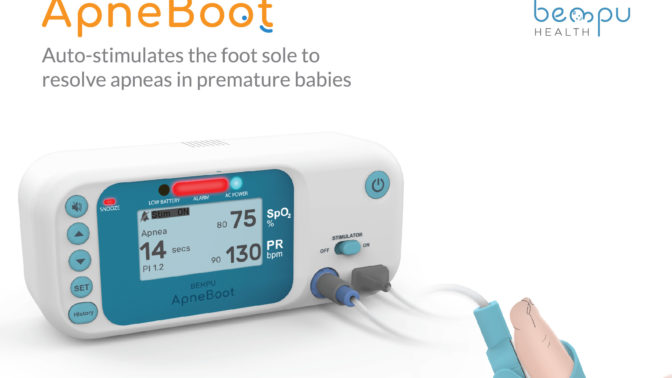
- ApneBoot has a built-in pulse oximeter with an alarm and auto-stimulation mechanism that fits on the newborn foot.
- The device has a built-in algorithm which monitors for drops in oxygen saturation and/or heart rate.
- In the event of an episode, ApneBoot vibrates against the newborn’s foot to stimulate the nervous system and restart breathing.
- The device also emits an audiovisual alarm to get the nurse’s attention in the case apnea is secondary and requires interventions.
- The device has battery back-up to last for 12 hours without recharge, providing continuous monitoring even under conditions of unreliable power supply or transport.
Key Features
BEMPU Health has developed ApneBoot, a wearable device to alert for bradycardia and hypoxia, key symptoms of neonatal apnea. Further, ApneBoot resolves episodes of apnea and prolonged hypoxia episodes in premature newborns through an instantaneous tactile stimulation to the foot sole. The device has a built-in pulse oximeter with an alarm and auto-stimulation mechanism that fits on the newborn foot in the convenient form factor of a baby boot. The device can be worn immediately upon birth and is designed to fit very small babies.
ApneBoot has a built-in algorithm which monitors for drops in oxygen saturation and/or heart rate. In the event of an episode, ApneBoot:
-
Vibrates against the newborn’s foot sole, which contains significant nerve endings. This stimulates the nervous system and restarts breathing.
-
Emits an audiovisual alarm to get the nurse’s attention in the case apnea is secondary and requires interventions more than stimulation like assisted ventilation for resolution.
The device also has battery back-up to last for 12 hours without recharge, providing continuous monitoring even under conditions of unreliable power supply or transport. It is portable, robust, reusable, and affordable making it an appropriate innovation for low-resource settings.
Social Impact
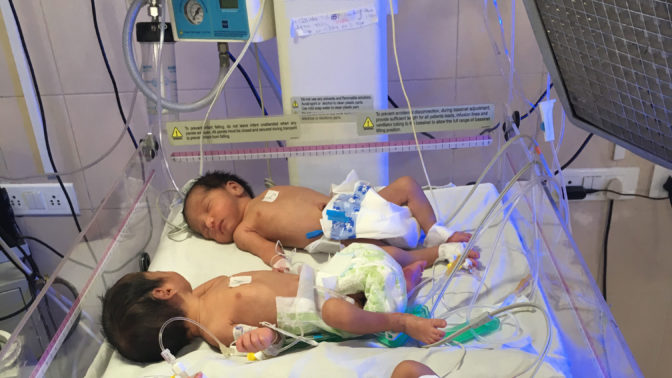
Apnea of Prematurity (AOP), a cessation of breathing is a serious risk factor for premature newborns. If unresolved immediately, AOP can cause severe injury and death. 3.3 million premature newborns in India and 15 million globally are at risk of apneas every year.
Apnea requires rapid intervention, but is compromised in low-resource settings, due to lack of vital sign monitors and skilled staff to respond to apneas (55% of Indian SNCUs have inadequate nursing staff). As a result, newborns in these facilities suffer from injury or death due to apnea and associated hypoxia.
To address this gap, we developed ApneBoot, a device to not only detect but also provide intervention to resolve AOP.
Our theory of change is that in premature newborns of resource-constrained settings, ApneBoot will terminate apneic and hypoxic episodes early, saving them from apnea and prolonged hypoxia-related injury and death, resulting in improved neurodevelopmental outcomes. The device has helped more than 120 newborns by providing seamless apnea monitoring, uses a high accuracy FDA, CE certified built-in pulse oximeter and has two journal publications.
Future Plans
We aim to deploy ApneBoot in all low- and middle-income countries, to strengthen their neonatal standard of care in NICUs. In the next 5 years, we will deploy approximately 5,200 devices in public and private health settings of India and LMICs, allowing us to impact around 67,600 premature newborns cumulatively.
We are also developing added versions of the device with new features driven by customer feedback.
Main Target Group
Hospitals and neonatologists caring for premature newborns in NICUs.
Main User
Premature newborns at risk of apneas.